 重要提示:
请勿将账号共享给其他人使用,违者账号将被封禁!
重要提示:
请勿将账号共享给其他人使用,违者账号将被封禁!
 题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
 更多“可逆热机的工作介质为理想气体,是一种理想的热机,实际不存在。”相关的问题
更多“可逆热机的工作介质为理想气体,是一种理想的热机,实际不存在。”相关的问题
第1题
一可逆热机用某种比热容不变的理想气体为工质,按下述循环操作,自状态1恒容加热到状态由状态2绝热膨胀到状态3,然后由状态3经恒压过程回到状态l,证明此循环中净功与所吸热量之为![]() ,其中γ=cp/cV。
,其中γ=cp/cV。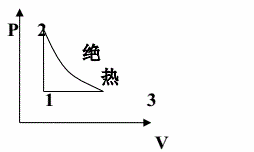
第2题
A. 甲、乙、丙、丁全对
B. 甲、乙、丙、丁全错
C. 甲、乙、丁对,丙错
D. 乙、丁对,甲、丙错.
第6题
理想气体是理论上假想的一种把实际气体性质加以简化的气体。人们把假想的,在任何情况下都严格遵守气体三定律的气体称为理想气体。就是说:一切实际气体并不严格遵循这些定律,只有在温度较高,压强不大时,偏离才不显著。所以一般可认为温度不低于0℃,压强不高于1.01×105Pa时的气体为理想气体。An ideal gas is a theoretical hypothetical gas which simplifies the properties of an actual gas. A hypothetical gas that strictly obeys the three laws of gas in all circumstances is called an ideal gas. That is to say, all real gases do not strictly follow these laws, and only at high temperatures and low pressures does the deviation become insignificant. Therefore, it can be generally regarded as an ideal gas when the temperature is not lower than 0℃ and the pressure is not higher than 1.01×105Pa.
第8题
一容器中装有1mol的单原子理想气体,温度为T1=546K,容器器壁的热容量可忽略不计。一循环热机从容器内的气体中吸热做功,并向一个温度为T2=273K的低温热源放热(设低温热源足够大,温度可近似看作不变),试求该热机最多能做多少功?

 警告:系统检测到您的账号存在安全风险
警告:系统检测到您的账号存在安全风险
为了保护您的账号安全,请在“上学吧”公众号进行验证,点击“官网服务”-“账号验证”后输入验证码“”完成验证,验证成功后方可继续查看答案!
